A Hipaa Authorization Has Which of the Following Charac
Read on to see what those items include. By signing the authorization an individual is giving consent to have their health information used.

Sec Filing F Star Therapeutics Inc
Authorization unless treatment is sought only to create health information for a third party or to take part in a research study and that I may have the right to refuse to sign this authorization.
. Cannot be combined with any other document related to the research. Valid a HIPAA authorization must satisfy the following 2. The authorization must be written in plain language.
HIPAA Checklist for a Valid Authorization 164508c 1 defines the following core elements for an authorization to disclose protected health information PHI. A HIPAA authorization has which of the following characteristics. Does the HIPAA Privacy Rule require documentation of Institutional Review Board IRB or Privacy Board approval of an alteration or waiver of individual authorization before a covered entity may use or disclose protected health information for any of the following provisions.
BCannot be combined with any other document related to the research. 2 Signed copy of Authorization is provided to individual signing it in case if entity covered is seeking Authorization. A copy of this authorization is as valid as the original.
A description of the specific information to be used or disclosed. The law requires that a HIPAA authorization form contain specific core elements to be valid. Uses plain language that the data subject can understand similar to the requirement for an informed consent document.
The answer is. Section 164508 c 1 i. I will receive a copy of this authorization after I have signed it.
The authorization may not be combined with any other document such as a consent for treatment3 An authorization to use or disclose psychotherapy notes may not be combined with an authorization to disclose other forms of PHI. In order to be accepted by doctors and hospitals a HIPAA release authorization must have six core requirements. Authorizations should have certain elements to be considered valid.
Some of this is used to file with the insurance company and it is also following HIPAA rules of privacy. The information is requested by a family member c. A specific and meaningful description of the PHI to be used or disclosed The identification of the persons or class of persons authorized to make the.
A HIPAA authorization has which of the following characteristics. If a covered entity seeks an authorization from an individual for a use or disclosure of protected health information the covered entity must provide the individual with a copy of the signed authorization. If youre unsure about the particulars of HIPAA research requirements at your organization or have questions you can usually consult with.
Has published the following guidelines that outline the differences between permitted HIPAA disclosures through an authorization and required disclosure under HIPAA right of access. A HIPAA authorization is a detailed document in which specific uses and disclosures of protected health are explained in full. The name or other specific identification of the person s or class of persons authorized to make the requested use or disclosure.
Therefore HIPAA rules of privacy protect the patient. Download our Free HIPAA Project Plan. A description that identifies the requested information in a specific and meaningful fashion 45 CFR.
A HIPAA authorization has which of the following characteristics. The individual who is subject of the information or the individuals personal representative authorizes in writing. A HIPAA authorization has which of the following characteristics.
A description of the information to be used or disclosed that identifies the information in a specific and meaningful fashion. 1 It must jotted down in simple language. If patient requests for records and release of information falls within your job.
Thus revocation of an authorization limits a covered entitys own continued use of the health. The Authorization has the following characteristics. Cannot be revoked by the data subject.
The patient must give authority for any. Valid HIPAA Authorization Requirements. A signed form gives your organization permission to use the patients PHI or disclose it to another person or entity.
3 Authorization is not considered valid until and unless it contains the required statements and elements. An authorization is a clients signed permission to allow a legal professional to obtain the clients PHI from a covered entity. The authorization may not be combined with any other document such as a consent for treatment.
To be valid a HIPAA authorization must satisfy the following 2. The authorization must be written in plain language not legalese. A valid authorization must contain certain required statements.
In case it is needed the authorization also gives the doctor permission for future medical treatment. A HIPAA authorization has which of the following characteristics. 1 for preparatory research at 45 CFR 164512i1ii 2for research on the protected health.
CUses plain language that the data subject can understand similar to the requirement for an informed consent document. Uses plain language that the data subject can understand similar to the requirement for an informed consent document. ACannot be revoked by the data subject.
A HIPAA authorization has which of the following characteristics. No one wants their personal information shared with just anyone. Under HIPAA retrospective research aka data mining on collections of PHI generally.
Uses plain language that the data subject can understand similar to the requirement for an informed consent document. 3 An authorization to use or disclose psychotherapy notes may not be combined with an authorization to disclose other forms of PHI. A valid HIPAA authorization must contain at least the following elements referred to as core elements.
164508 contains the core elements and required statements that must be included in a HIPAA compliance authorization. An authorization in HIPAA terms is the consent of an individual or patient providing explicit authorization to use or disclose their personal information. A HIPAA authorization can allow a covered entity to use or disclose an individuals PHI for its own research purposes or disclose PHI to another entity for that entitys research activities.
A HIPAA Authorization has Which of The Following CharacteristicsRecruiting Into ResearchUnder HIPAARetrospective ResearchInstitution COI Quizlet. 4 Copy to the individual. Uses plain language that the data subject can understand similar to the requirement for an informed consent document.
5The covered entity may use or disclose protected health information when. A HIPAA authorization form represents an agreement between a patient and a HIPAA-covered organization.

A Hipaa Authorization Has Which Of The Following Characteristics Seoblogger7 Com
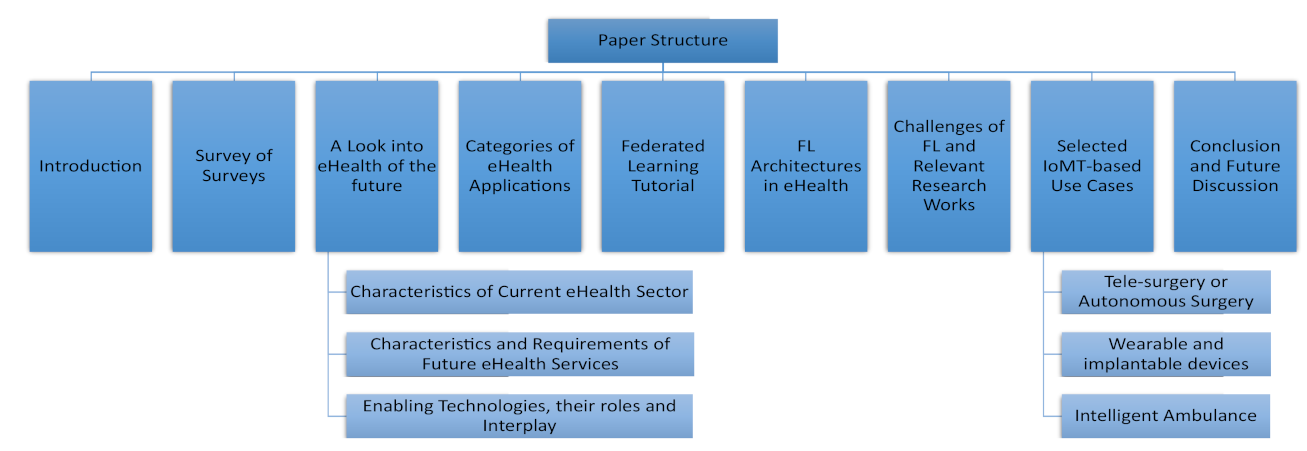
Sensors Free Full Text Rebirth Of Distributed Ai A Review Of Ehealth Research Html
%20(27).png)
A Hipaa Authorization Has Which Of The Following Characteristics Seoblogger7 Com

Protected Health Information Hipaa Uci Office Of Research

What Is Phi Protected Personal Health Information

Research And Hippa Privacy Citi Docx Question 1 Under Hipaa A U201cdisclosure Accounting U201d Is Required For All Research Where The Data Crosses State Course Hero

Research And Hipaa Privacy Protections Quizzma

Research And Hipaa Privacy Protections Id 14 Flashcards Quizlet

Research And Hipaa Privacy Protections Quizzma

Researching The Appropriateness Of Care In The Complementary And Integrative Health Professions Part 2 What Every Researcher And Practitioner Should Know About The Health Insurance Portability And Accountability Act And Practice Based Research




Comments
Post a Comment